THE R22 ROLLOUT:
END OF MALARIA?
The 2026 Scaling Strategy
Local Manufacturing
By 2026, tech transfer agreements have allowed Ghana, Nigeria, and India to produce the vaccine locally. This has slashed costs and removed the vulnerability of international shipping delays.
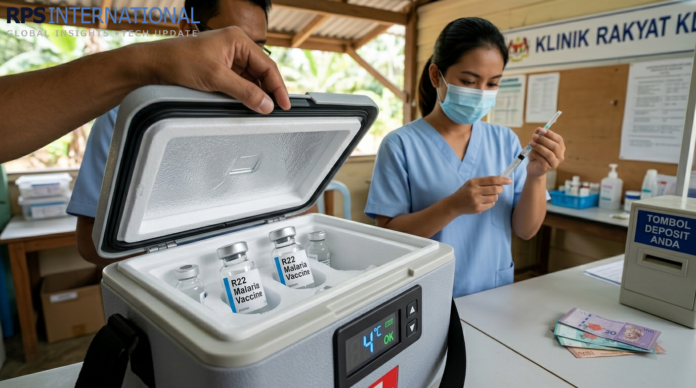
Standard Cold Chain
Unlike mRNA vaccines that require ultra-cold storage, R22 remains stable in a standard 2-8°C refrigerator. This allows for deep penetration into rural districts where advanced infrastructure is absent.
“The R22 vaccine is not just a medical triumph; it is a logistical one. For the first time, we have a highly effective vaccine that is cheap enough and stable enough to reach every child, in every village, across the entire African continent.”
— Dr. Amara Okafor, Global Health Initiative 2026
A Future Without Malaria
Support the global rollout and stay updated on distribution progress in your region.