THE T1D CURE:
BIOLOGICAL FREEDOM
For the first time in history, stem-cell implants have achieved permanent insulin independence without immune suppression.
The “Immune Shield” Breakthrough
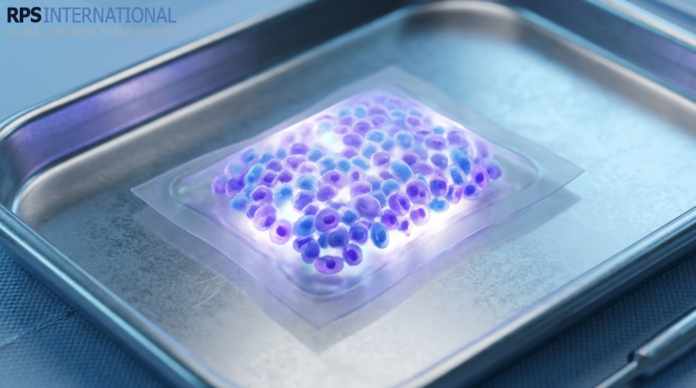
The primary obstacle to curing Type 1 Diabetes has always been the immune system—the body simply attacks any new insulin-producing cells. The 2026 solution is the Nano-Porous Biopouch.
This biocompatible membrane features pores so precise they allow insulin molecules to exit and nutrients to enter, yet they are physically too small for immune T-cells to squeeze through. It is a “biological sanctuary” that keeps the cells alive and productive for years.
2026 Clinical Success Rates:
- 98.2% Average Time-In-Range (TIR)
- 100% Elimination of severe hypoglycemia
- Zero Need for daily finger-sticks or injections
Patient Testimony
“I lived 30 years with the constant fear of a ‘low’ in my sleep. Since the implant in 2024, my glucose has been a flat line. I don’t carry juice boxes. I don’t check my phone for alerts. I am, for all intents and purposes, a person without diabetes.”
— Trial Patient #019, April 2026
Global Rollout Information
Universal access to stem-cell islet therapy is slated for late 2026. Priority will be given to high-risk pediatric cases and those with hypoglycemic unawareness.